Molecule: Tislelizumab
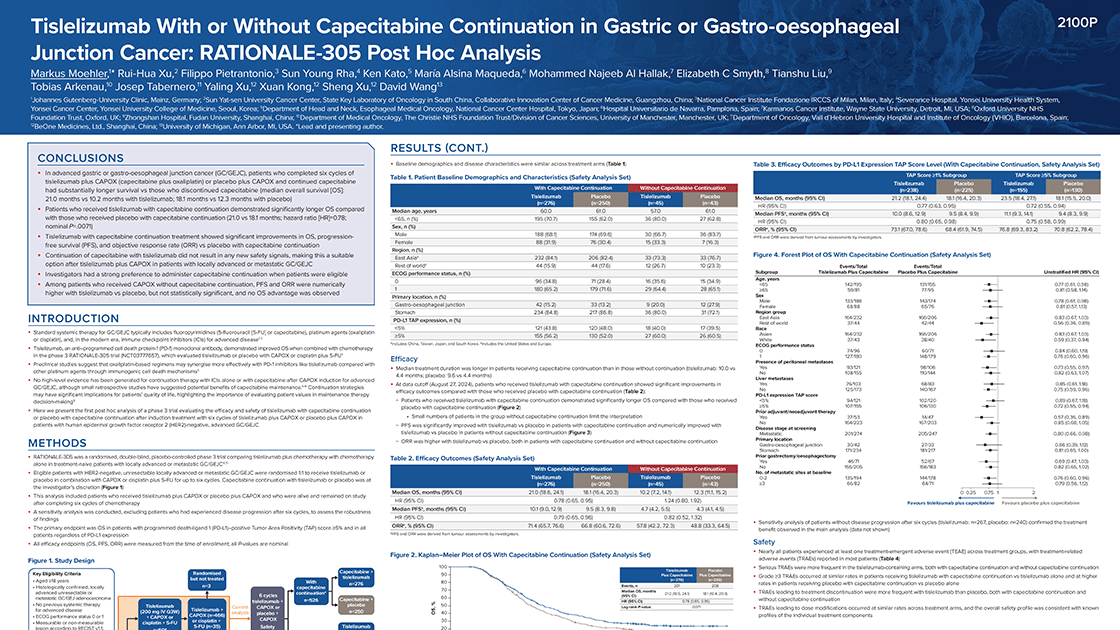
RATIONALE-305 (NCT03777657) is a randomized, double-blind, placebo-controlled, global Phase 3 study of tislelizumab combined with platinum and fluoropyrimidine chemotherapy compared to placebo combined with platinum and fluoropyrimidine chemotherapy as a first-line treatment for patients with advanced unresectable or metastatic gastric or gastroesophageal junction adenocarcinoma (GC/GEJC).