Disease State: Waldenström's macroglobulinemia
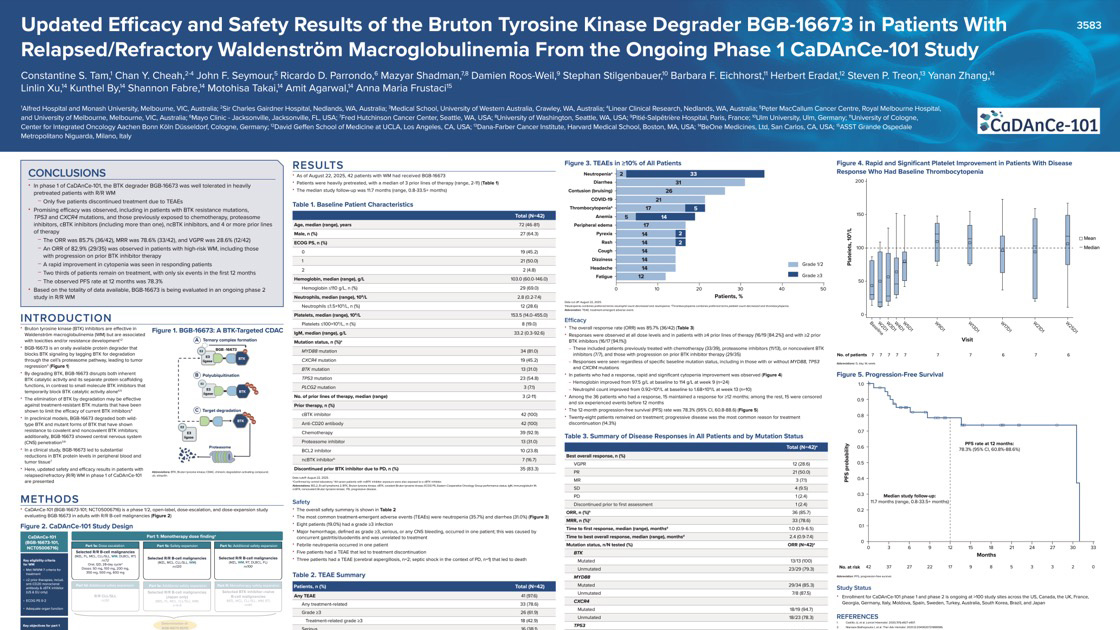
CaDAnCe-101 (NCT05006716) is a Phase 1/2, open-label, dose-escalation and expansion trial evaluating BGB-16673, a Bruton tyrosine kinase (BTK) degrader, in patients with relapsed/refractory B-cell malignancies, including chronic lymphocytic leukemia/small lymphocytic lymphoma (CLL/SLL), follicular lymphoma (FL), and marginal zone lymphoma (MZL).
CaDAnCe-101 (NCT05006716) is a Phase 1/2, open-label, dose-escalation and expansion trial evaluating BGB-16673, a Bruton tyrosine kinase (BTK) degrader, in patients with relapsed/refractory B-cell malignancies, including chronic lymphocytic leukemia/small lymphocytic lymphoma (CLL/SLL), follicular lymphoma (FL), and marginal zone lymphoma (MZL).