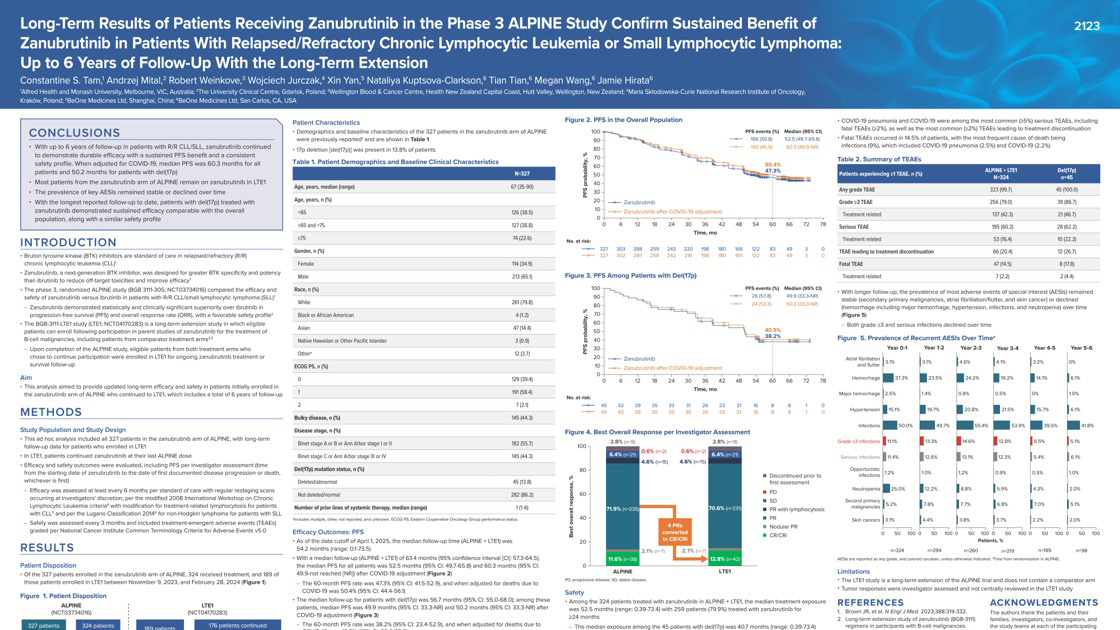
This report presents up to six years of follow-up for patients with relapsed/refractory chronic lymphocytic leukemia/small lymphocytic lymphoma (R/R CLL/SLL) treated with the BTK inhibitor zanubrutinib in the ALPINE study. Zanubrutinib continued to show durable benefits, with nearly half of patients remaining progression-free. Patients with high-risk genetic features (del(17p)) showed responses comparable to other patients in the study. Serious adverse events (AEs) were seen in 60% of patients, grade ≥3 AEs occurred in 79%, treatment-related grade ≥3 AEs in 42%, and 20% discontinued due to AEs. Fatal TEAEs occurred in 47 patients, most frequently due to infection. The safety profile remained consistent over time, with most side effects stable year-to-year. These long-term data support zanubrutinib as an effective treatment option for R/R CLL/SLL.
The contents of the presentations above are designed for educational and scientific exchange purposes and are not promotional. They may contain information on investigational products or investigational uses of approved products. No conclusions regarding safety and/or efficacy for such investigational products or uses may be made.