DISCLAIMER
The contents of the presentations above are designed for educational and scientific exchange purposes and are not promotional. They may contain information on investigational products or investigational uses of approved products. No conclusions regarding safety and/or efficacy for such investigational products or uses may be made.
SUMMARY
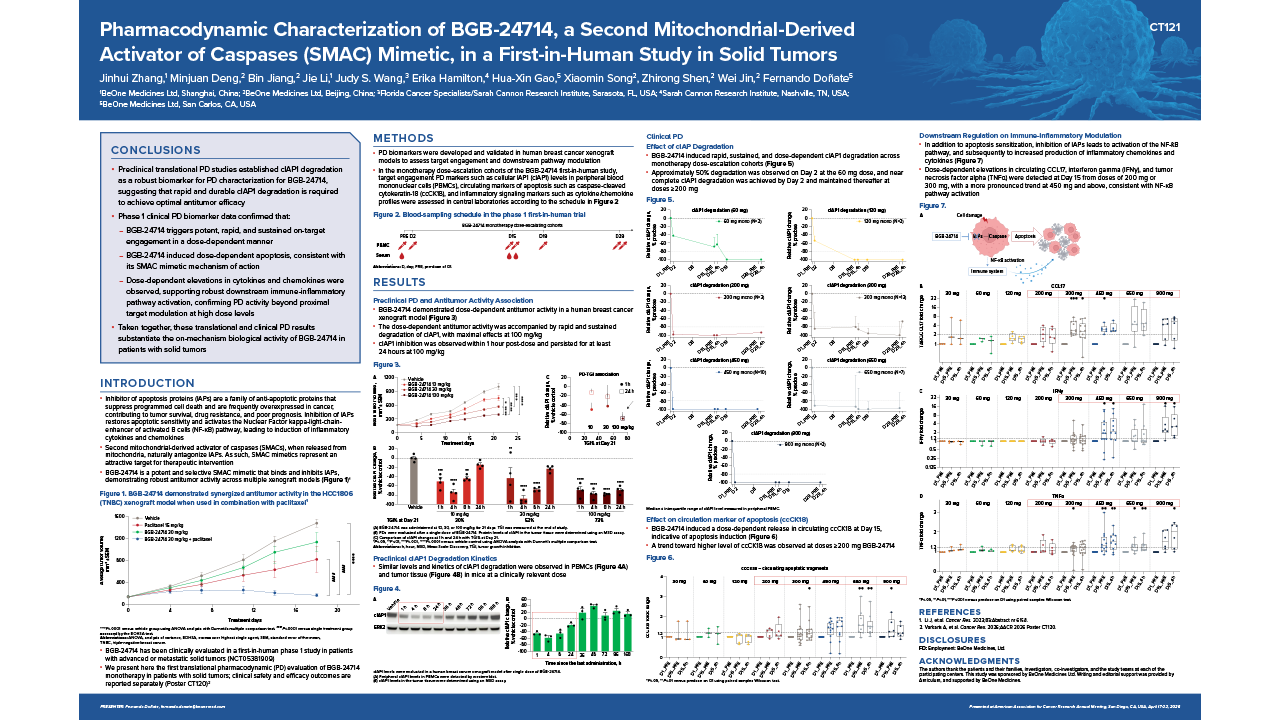
This presentation reported translational pharmacodynamic biomarker findings from a first-in-human, dose-escalation study of BGB-24714, an investigational, selective second mitochondrial-derived activator of caspases (SMAC) mimetic, in patients with solid tumors. In the monotherapy dose-escalation cohorts, cellular inhibitor of apoptosis protein 1 (cIAP1) levels in peripheral blood mononuclear cells were evaluated to assess target engagement, along with caspase-cleaved cytokeratin-18 (ccCK18) and circulating cytokines and chemokines profiles to measure apoptosis and inflammatory signaling, respectively. BGB-24714 demonstrated rapid, sustained, and dose-dependent cIAP1 degradation, achieving ~50.0% degradation by Day 2 at 60 mg and near-complete degradation by Day 2 at doses ≥200 mg. Furthermore, dose-dependent increases in ccCK18 and inflammatory cytokines and chemokines at Day 15 were noted at doses ≥200 mg, indicating apoptosis induction and NF-κB pathway activation.
ClinicalTrials.gov ID: NCT05381909
PICO TABLE
| Population | Intervention | Comparator | Outcome Measures |
|---|---|---|---|
| Patients with solid tumors | BGB-24714 | N/A |
|
FREQUENTLY ASKED QUESTIONS
The objective was to characterize pharmacodynamic biomarkers reflecting target engagement and downstream pathway modulation of BGB-24714, an investigational, selective second mitochondrial-derived activator of caspases (SMAC) mimetic, in patients with solid tumors.
Pharmacodynamic biomarkers were developed and validated in preclinical models prior to clinical application. Evaluation was conducted in monotherapy dose-escalation cohorts; blood samples were collected pre-dose (baseline) and at serial post-dose timepoints. Cellular inhibitor of apoptosis protein 1 (cIAP1) levels in peripheral blood mononuclear cells was quantified to assess target engagement. Caspase-cleaved cytokeratin-18 (ccCK18) was measured as a circulating marker of apoptosis, and cytokine and chemokine profiles were analyzed to evaluate activation of inflammatory signaling pathways, including nuclear factor kappa B (NF-κB) signaling.
BGB-24714 induced rapid, sustained, and dose-dependent cIAP1 degradation in peripheral blood mononuclear cells. Approximately 50.0% degradation was observed by Day 2 at the 60 mg dose, while near-complete degradation by Day 2 was reported at doses ≥200 mg and maintained thereafter. Dose-dependent increases in ccCK18 and inflammatory cytokines and chemokines at Day 15 were noted at doses ≥200 mg, indicating apoptosis induction and NF-κB pathway activation.